2006 – 2008
Removal of Critical Contaminants from Sewage Water by Means of Membrane Filtration and Photocatalysis (Willy Hager III)
| Contact: | |||
|---|---|---|---|
| Project Group: | Project | ||
| Funding: | Willy-Hager-Stiftung | ||
Period
Description
Removal of Critical Contaminants from Sewage Water by Means of Membrane Filtration and Photocatalysis
(WILLY-HAGER III)
One disadvantage of photocatalytic oxidation with TiO2 in water purification is the fact that for efficient water purification a large irradiated surface is needed, which must be also accessible for the target contaminants. Due to the surface properties of TiO2 the relevant substances (e.g. pharmaceuticals, x-ray contrast media), which are mainly nonpolar, offer poor absorbance behaviour. As the photocatalytic degradation takes place either directly at the TiO2 surface or in direct proximity via OH radicals with short lifetimes, the low adsorption is the limiting step.
The application of activated charcoal (AC) can be an efficient means to reduce the concentration of nonpolar substances but a stationary operation mode is not possible, since the loaded AC usually needs to be regenerated externally.
The combination of TiO2/AC could offer a solution for both problems (link). The attendance of AC within photocatalytic processes can lead to an increased degradation of the target contaminants. Furthermore a regeneration and/or a circuitry of the AC and thus a stationary operation mode is possible. The composition of the suspension can be chosen flexibly and existing plants for the photodegradation with pure TiO2 suspensions can be used without modifications.
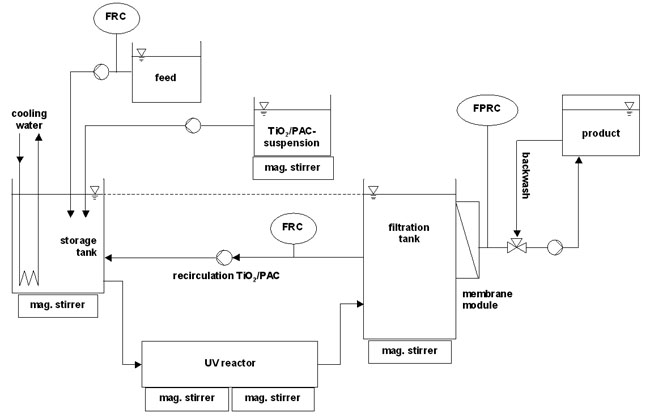
Figure 1: Flow scheme of the experimental setup for the combination of photocatalysis and membrane filtration
In lab scale experiments concerning the degradation with different pharmaceuticals could be shown that, depending on the used system, a significant synergistic effect arises (1.5 times higher degradation rates for clofibric acid) which can be used for an accelerated photocatalytic degradation. Besides by addition of AC the filtration of the TiO2 suspension can be conducted more efficiently. A partial regeneration of the AC by photocatalytically generated OH radicals is possible, but it needs to be proofed in longterm experiments. In further experiments a pilot plant is installed to transfer the results from the preliminary tests to a larger scale.

